Checkmate 9la brain metastases8/31/2023 Fatal adverse reactions occurred in 1% of patients these included events of pneumonitis (0.6%). The most frequent serious adverse reaction reported in ≥2% of patients receiving OPDIVO was urinary tract infection. In Checkmate 274, serious adverse reactions occurred in 30% of patients receiving OPDIVO (n=351). The most frequent serious adverse reactions reported in ≥2% of patients receiving OPDIVO were urinary tract infection, sepsis, diarrhea, small intestine obstruction, and general physical health deterioration. In Checkmate 275, serious adverse reactions occurred in 54% of patients receiving OPDIVO (n=270). The most frequent serious adverse reactions reported in ≥2% of patients receiving OPDIVO were pneumonia, dyspnea, respiratory failure, respiratory tract infection, and sepsis. In Checkmate 141, serious adverse reactions occurred in 49% of patients receiving OPDIVO (n=236). Eleven patients died from causes other than disease progression: 3 from adverse reactions within 30 days of the last OPDIVO dose, 2 from infection 8 to 9 months after completing OPDIVO, and 6 from complications of allogeneic HSCT. The most frequent serious adverse reactions reported in ≥1% of patients were pneumonia, infusion-related reaction, pyrexia, colitis or diarrhea, pleural effusion, pneumonitis, and rash. Serious adverse reactions occurred in 26% of patients. In Checkmate 205 and 039, adverse reactions leading to discontinuation occurred in 7% and dose delays due to adverse reactions occurred in 34% of patients (n=266). The most frequent serious adverse reactions reported in ≥2% of patients were acute kidney injury, pleural effusion, pneumonia, diarrhea, and hypercalcemia. In Checkmate 025, serious adverse reactions occurred in 47% of patients receiving OPDIVO (n=406). Fatal intestinal perforations occurred in 3 (0.9%) patients. The most frequent serious adverse reactions reported in ≥2% of patients were diarrhea, pneumonia, pneumonitis, pulmonary embolism, urinary tract infection, and hyponatremia. In Checkmate 9ER, serious adverse reactions occurred in 48% of patients receiving OPDIVO and cabozantinib (n=320). The most frequent serious adverse reactions reported in ≥2% of patients were diarrhea, pyrexia, pneumonia, pneumonitis, hypophysitis, acute kidney injury, dyspnea, adrenal insufficiency, and colitis. 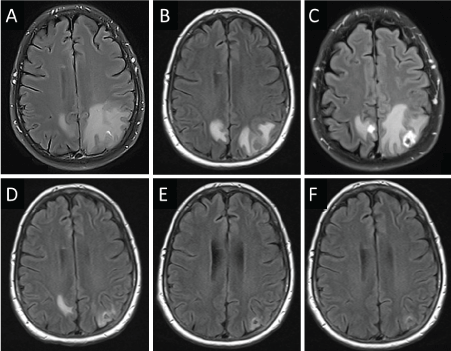
In Checkmate 214, serious adverse reactions occurred in 59% of patients receiving OPDIVO plus YERVOY (n=547). Fatal adverse reactions occurred in 4 (1.3%) patients and included pneumonitis, acute heart failure, sepsis, and encephalitis. The most frequent serious adverse reactions reported in ≥2% of patients were pneumonia, pyrexia, diarrhea, pneumonitis, pleural effusion, dyspnea, acute kidney injury, infusion-related reaction, musculoskeletal pain, and pulmonary embolism. In Checkmate 743, serious adverse reactions occurred in 54% of patients receiving OPDIVO plus YERVOY. In Checkmate 057, fatal adverse reactions occurred these included events of infection (7 patients, including one case of Pneumocystis jirovecii pneumonia), pulmonary embolism (4 patients), and limbic encephalitis (1 patient). The most frequent serious adverse reactions reported in ≥2% of patients receiving OPDIVO were pneumonia, pulmonary embolism, dyspnea, pyrexia, pleural effusion, pneumonitis, and respiratory failure. In Checkmate 017 and 057, serious adverse reactions occurred in 46% of patients receiving OPDIVO (n=418). Fatal adverse reactions occurred in 7 (2%) patients, and included hepatic toxicity, acute renal failure, sepsis, pneumonitis, diarrhea with hypokalemia, and massive hemoptysis in the setting of thrombocytopenia. The most frequent (>2%) serious adverse reactions were pneumonia, diarrhea, febrile neutropenia, anemia, acute kidney injury, musculoskeletal pain, dyspnea, pneumonitis, and respiratory failure. 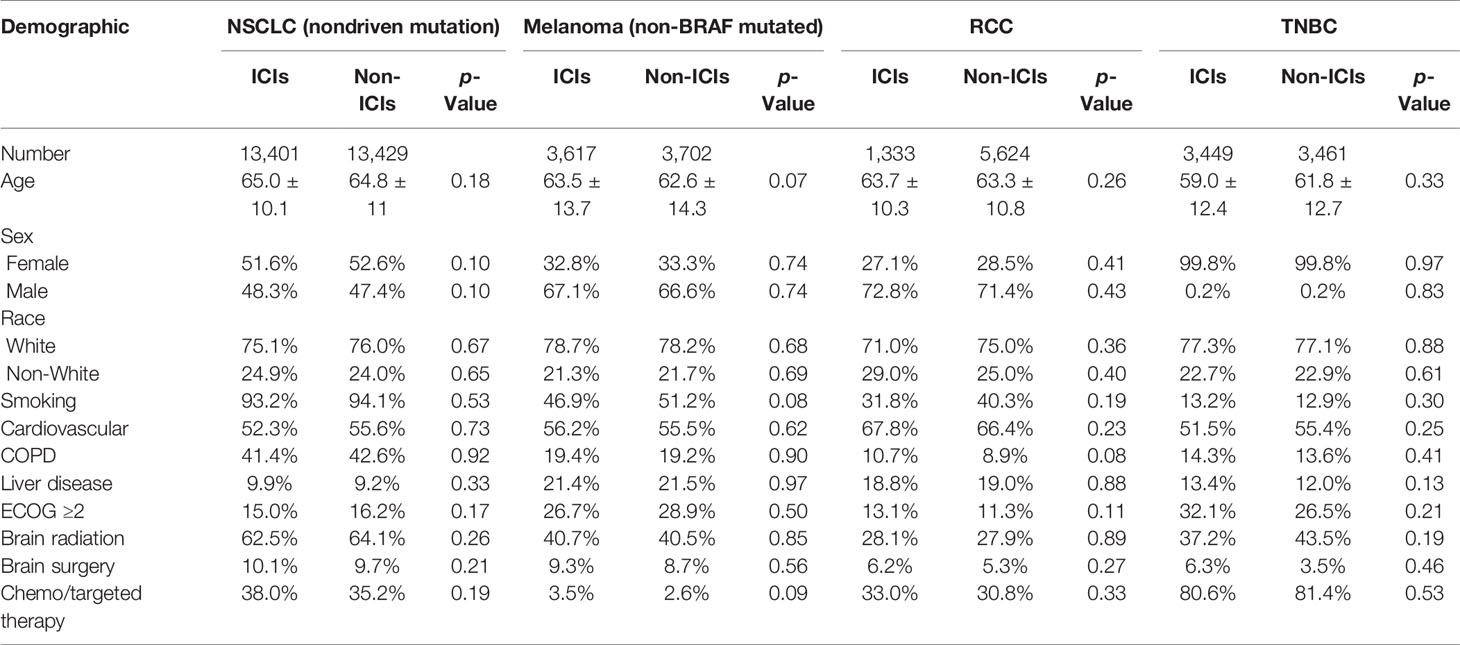
In Checkmate 9LA, serious adverse reactions occurred in 57% of patients (n=358). Fatal adverse reactions occurred in 1.7% of patients these included events of pneumonitis (4 patients), myocarditis, acute kidney injury, shock, hyperglycemia, multi-system organ failure, and renal failure. The most frequent (≥2%) serious adverse reactions were pneumonia, diarrhea/colitis, pneumonitis, hepatitis, pulmonary embolism, adrenal insufficiency, and hypophysitis. In Checkmate 227, serious adverse reactions occurred in 58% of patients (n=576). No fatal adverse reactions occurred in patients who received OPDIVO in combination with platinum-doublet chemotherapy. Dual immunotherapy-based combination resulted in clinical benefits across key subgroups of mNSCLC patients with high unmet need, including those with PD-L1 expression 2% included pneumonia and vomiting.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
